Pipeline
CU103
Pipeline
CU103
CU103
Expanded indication of CU06 for Stroke
| Sortation | content |
|---|---|
| CU103 | Expanded indication of CU06 for Stroke |
| Indication | Stroke |
| Unmet Needs |
• A leading cause of death and disability globally, particularly in low-income and middle-income countries, and this burden is increasing. • Estimated prevalence of unmet needs was 59.7% to 83.7% for body functioning, and 48.4% for activities/participation. (source: BMJ 2019) • Urgent need to develop effective neuroprotective agents for the prevention and treatment of cerebral ischemia |
| Mechanism of Action |
• Preserves BBB integrity by stabilizing the EC junction and reduces astrocyte end-feet swelling following I/R. • CU103 attenuates inflammation responses by inhibiting NF-κB activation • CU103 protects the brain from I/R injury by preventing BBB disruption and inflammation |
| Efficacy & Safety |
• Reduction of infarction volume and edema in a cerebral I/R mouse model • Inhibition of astrocyte end-feet swelling and activation after I/R • Prevention of disruption of basal membrane proteins in endothelial cells after I/R • Decrease in of cerebral injury after I/R in rat model |
| Market |
• 5.1 billion US$ in 2021 (8 Major Markets) for Acute Ischemic Stroke • Forecasted to be 7.3 billion US$ in 2027 (8 Major Markets) for Acute Ischemic Stroke |
Indication
Cause
Incidence, Mortality
Symptoms
Unmet Needs
Mechanism of Action
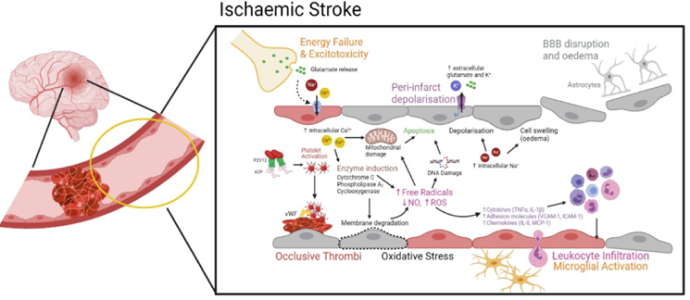
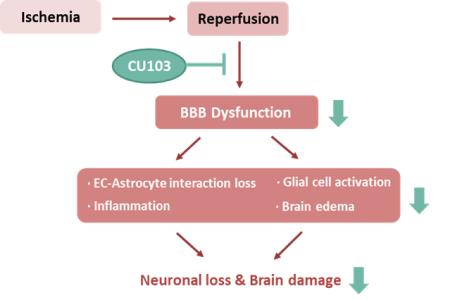
The CU103, EC dysfunction blocker has neuro-protective activities by maintaining BBB integrity, demonstrates a promising drug for stroke
Efficacy
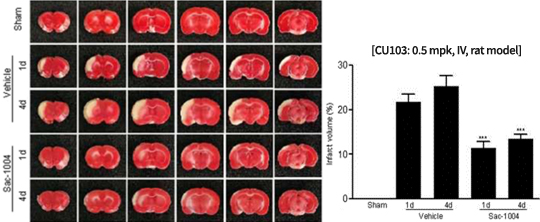
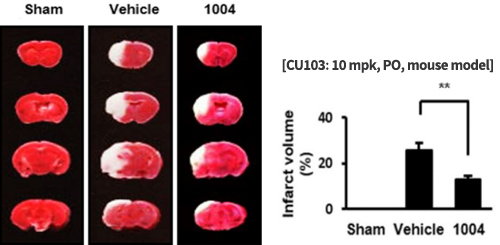
Reduction of infarction volume and edema in cerebral I/R mouse model
CU103 attenuates infarction region in MCAO mouse model
I/R: Ischemia-Reperfusion
MCAO: Middle Cerebral Artery Occlusion
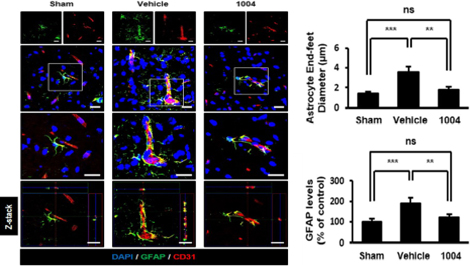
Inhibition of astrocyte end-feet swelling and activation after I/R
CU103 likely alleviates astrocyte end-feet destruction by stabilizing the vascular structure after I/R
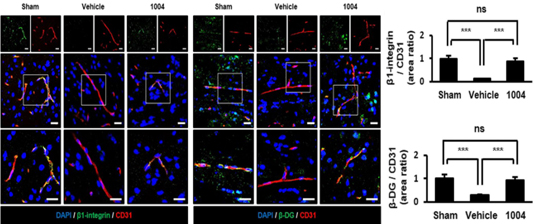
Prevention of disruption of basal membrane proteins in endothelial cells after I/R
CU103 recovers β1-integrin & β-DG expression
Decrease in cerebral injury after I/R in rat model
Similar effect confirmed in the rat (larger animal) model and mouse model